-
 play_arrow
play_arrow
BayRadio Listen Live Broadcasting in Spain
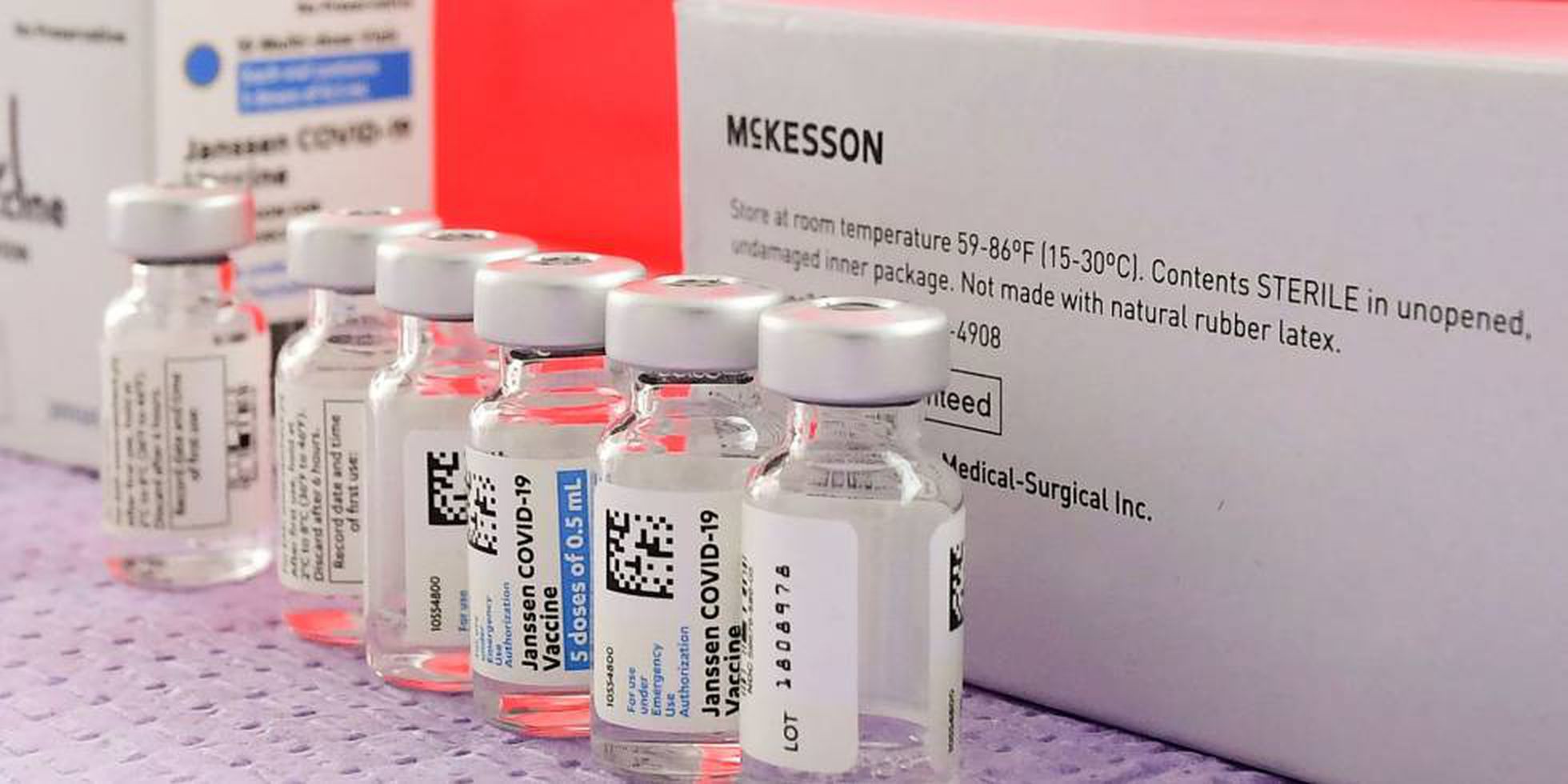
THE acceleration of the Covid-19 vaccination programme across Spain may be hit by American health regulators putting the single-shot Janssen vaccine on hold for safety checks.
Pharmaceutical company Johnson and Johnson suspended its rollout while six cases of rare blood clots are investigated after seven million people received the jab in the United States.
The decision has a knock-on effect for Spain which was scheduled to start using the Janssen shot from today – another blow for the troubled immunisation campaign across Europe which was due to receive 5.5 million doses between April and June.
Madrid says the target of vaccinating 70% of the Spanish population this summer can still be reached but Janssen was going to be a key factor in its programme.
A total of 300,000 doses were arriving this month but over the next quarter, Spain was expecting to receive 5.5 million Janssen jabs and problems could occur should the suspension drag on – putting more pressure on Pfizer and Moderna fulfilling their own delivery targets.
José Antonio Forcada of the National Association of Nursing and Vaccines said: “If this is a stoppage of a few days, it won’t be a problem; but if it drags on, the acceleration of the speed that we were expecting won’t happen.”
Written by: BayRadio News
Similar posts
Recent Posts
- Sports injuries on the rise among people who start training without prior preparation
- A benchmark in Alicante for complex patients: How the Internal Medicine Service at Quirónsalud Torrevieja works
- Quirónsalud Torrevieja strengthens its Cardiology Service as a benchmark in advanced cardiovascular diagnosis and treatment
- An accurate diagnosis: the key to properly treating Irritable Bowel Syndrome and SIBO
- Marblanc Solar Completes First Solar Installation on Spain’s Costa Blanca

Ctra. Cabo La Nao, CC La Nao, Local 6 03730 Javea, Alicante, Spain
Advertise with us
Do you have a business in Spain? Do you provide a service to the expat community in Spain? Would you like your message to reach over 500.000 people on a weekly basis?
BayRadio is a community orientated radio station offering fantastic content to our many listeners and followers across our various platforms. Contact us now and find out what Bay can do for you!
Our business is helping your business grow.
BAY RADIO S.L. © 2024. ALL RIGHTS RESERVED. WEB DESIGN BY MEDIANIC