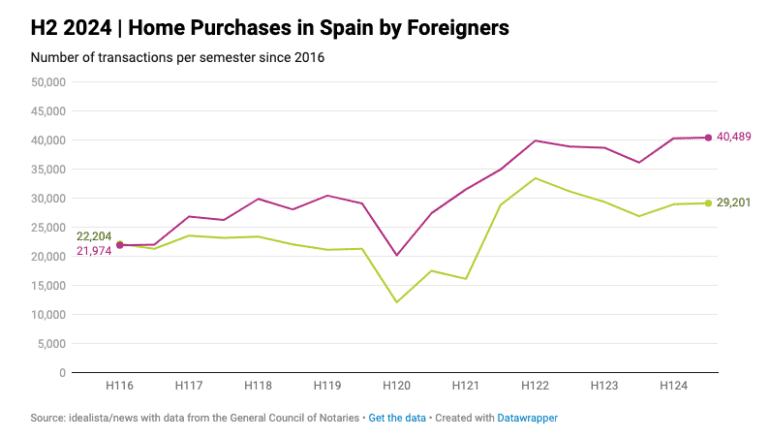
-
 play_arrow
play_arrow
BayRadio Listen Live Broadcasting in Spain
Communities continue to await EMA decision on third dose vaccine

The Autonomous Communities are awaiting the decision of the EMA (the European Medicines Agency) on the suitability of injecting a third dose of the coronavirus vaccine to the entire population that already has both doses.
This agency is expected to make its decision in the coming days. Views on whether or not it should be applied vary widely across countries. In Spain, for example, the Minister of Health, Carolina Darias, said in July that “everything points to yes,” while the president of the Spanish Vaccine Agency, Amós García Rojas, said that there is still no scientific evidence to support this decision.
The EMA meeting was scheduled for the afternoon of this Monday, 23 August, but it has been postponed and the decision will be taken in the coming days.
In this situation, the Community of Madrid is considering the possibility of inoculating this third dose of the coronavirus vaccine to immunocompromised or vulnerable people over the age of 65.
This has been assured by the Health Councillor of the Community of Madrid, Enrique Ruiz Escudero, who has stated that it is necessary to rely on “scientific and technical studies that support the possibility of administering this third dose and that it will generate a higher degree of protection”.
In the absence of a final decision by the EMA, the World Health Organisation (WHO) and the European Centre for Disease Control have taken a position in this debate, the regions of Spain hope to take a joint decision, as the communities have said.
While waiting for the meeting and final decision, Spain has received this Monday more than 2.3 million doses of the Pfizer vaccine against coronavirus, which has already begun to be distributed among the territories.
The new shipment of Pfizer is added to the 47,452,659 doses already delivered by this laboratory since the beginning of the vaccination campaign. In total, more than 63 million doses of the 68.3 of the four vaccines authorised in Europe have been administered in Spain.
Written by: BayRadio News
Similar posts
Recent Posts
- Alicante Boat Tours: Enjoy the Mediterranean Sea
- Avoiding extreme heat, staying well hydrated and consulting a specialist: key measures for controlling headaches
- Nina, a student at Lady Elizabeth School, invited to share her ideas about the future at TEDxThe British School of Málaga Youth
- Robotic surgery reduces the surgical impact on patients with ovarian cancer
- Sports injuries on the rise among people who start training without prior preparation

Ctra. Cabo La Nao, CC La Nao, Local 6 03730 Javea, Alicante, Spain
Advertise with us
Do you have a business in Spain? Do you provide a service to the expat community in Spain? Would you like your message to reach over 500.000 people on a weekly basis?
BayRadio is a community orientated radio station offering fantastic content to our many listeners and followers across our various platforms. Contact us now and find out what Bay can do for you!
Our business is helping your business grow.
BAY RADIO S.L. © 2024. ALL RIGHTS RESERVED. WEB DESIGN BY MEDIANIC