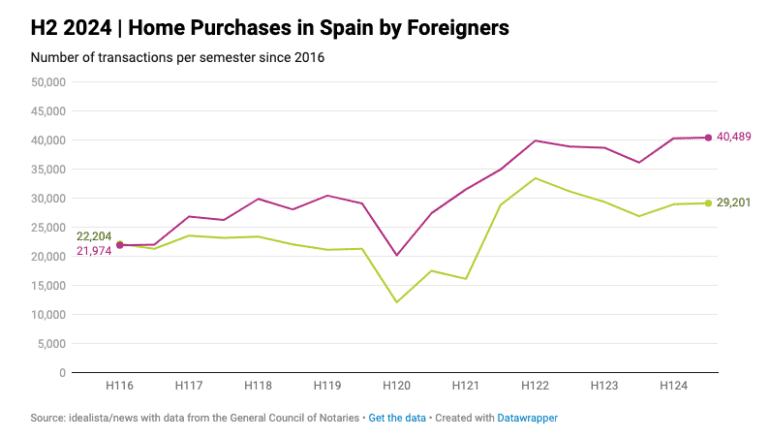
-
 play_arrow
play_arrow
BayRadio Listen Live Broadcasting in Spain
A study finds a way to avoid chemotherapy in patients with breast cancer

Ramón y Cajal University Hospital and its Health Research Institute IRYCIS are participating in the study of the new therapeutic strategy that could prevent chemotherapy in early HER-2+ breast cancer. Specifically, Medsir, a global company dedicated to clinical research in oncology, presents the continuation of the clinical trial ‘PHERGain-II’.
This aims to demonstrate that in patients with HER2-positive early breast cancer with good prognosis, a chemotherapy-free therapeutic approach can be as effective as the current standard of care, which involves the use of this generally more toxic treatment.
According to the American Society of Clinical Oncology (ASCO), the purpose of PHERGain-II is aligned with one of the main priorities in cancer research: that clinical research in oncology prioritizes the reduction of toxicity associated with cancer treatment.
“Given the side effects and impact of chemotherapy on the quality of life of patients with early breast cancer, it is important to develop therapeutic de-escalation strategies that allow the elimination of traditional systemic chemotherapy in correctly selected patient profiles,” says Dr Alfonso Cortés, principal investigator of the study and medical oncologist at the Ramón y Cajal University Hospital/IRYCIS.
“This study could mean in the near future the end of chemotherapy in a significant percentage of patients with this type of tumor,” explains Doctor Cortés. Precisely, HER-2+ breast cancer is a type of cancer with high presence of the human epidemic growth factor receptor 2 (HER-2), a protein that promotes the growth of cancer cells.
When these tumors are found early, they are removed by surgery and treated with therapies aimed at blocking HER-2. However, even in patients with a good prognosis, combination with chemotherapy remains part of standard treatment to prevent tumor recurrence after surgery.
Also, the ‘PHERGain-II’ trial will give patients preoperative treatment with trastuzumab and pertuzumab, drugs aimed at blocking HER-2, without chemotherapy. Tumors will be scanned by MRI before and after treatment to see if it is possible to predict the level of pathological response achieved after treatment.
After surgery, treatment will be continued with the same combination of drugs or with T-DM1 (another HER-2 blocking drug), reserving the use of chemotherapy only to those patients who have responded worst to preoperative treatment and who have a worse prognosis.
Written by: BayRadio News
Similar posts
Recent Posts
- Alicante Boat Tours: Enjoy the Mediterranean Sea
- Avoiding extreme heat, staying well hydrated and consulting a specialist: key measures for controlling headaches
- Nina, a student at Lady Elizabeth School, invited to share her ideas about the future at TEDxThe British School of Málaga Youth
- Robotic surgery reduces the surgical impact on patients with ovarian cancer
- Sports injuries on the rise among people who start training without prior preparation

Ctra. Cabo La Nao, CC La Nao, Local 6 03730 Javea, Alicante, Spain
Advertise with us
Do you have a business in Spain? Do you provide a service to the expat community in Spain? Would you like your message to reach over 500.000 people on a weekly basis?
BayRadio is a community orientated radio station offering fantastic content to our many listeners and followers across our various platforms. Contact us now and find out what Bay can do for you!
Our business is helping your business grow.
BAY RADIO S.L. © 2024. ALL RIGHTS RESERVED. WEB DESIGN BY MEDIANIC